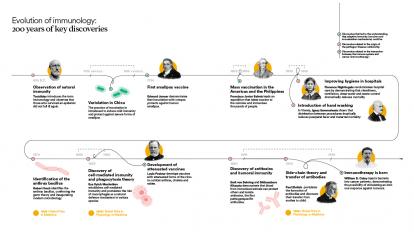
From the earliest observations that some survivors of the plague did not contract the disease again to the therapies that today modulate and reactivate our defences against cancer, immunology has completely transformed medicine. What began as intuitions about epidemics and vaccines is now a key science for understanding infections, tumours and autoimmunity, and for learning how to modulate our defences throughout life. How did it all begin, and what do we know about the immune system today?
For most of history, humanity fought diseases without understanding them. The first ideas that shaped immunology did not emerge from laboratories or complex theories, but from the direct observation of patients. One of the most decisive insights was the realisation that people who survived a particular epidemic rarely contracted the same disease again.
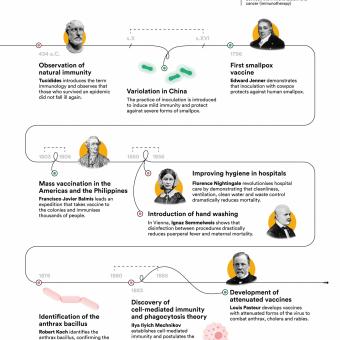
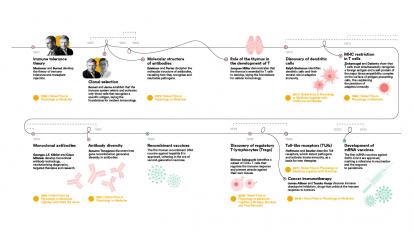
In 434 BC, during the Plague of Athens, the historian Thucydides described how survivors were protected from further infection by the disease, leaving a written record of a phenomenon that we now identify as acquired immunity. From this observation arose a powerful intuition: the body remembers diseases and learns how to attack them, allowing it to defend itself more efficiently in the future.
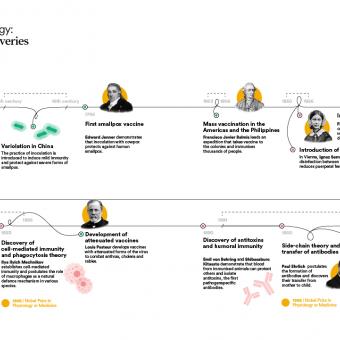
This intuition led people to search for ways of infecting healthy individuals in a controlled manner in order to stimulate this learning process in the body without exposing them to the full consequences of the disease. The question was not yet what caused illness, but how to achieve protection without dying in the process. There is evidence that as early as the 6th century BC, in China and India, the inoculation or nasal inhalation of powdered scabs from infected patients was practised as a means of preventing smallpox. Later, variolation was developed, a practice that involved deliberately inducing mild infections of smallpox to prevent its deadly forms. Healthy individuals were inoculated with material such as pus or scabs from patients with mild cases, producing an attenuated form of the disease and thereby generating immunity.
The vaccine: a revolution in public health
The intuitive search for protection through controlled exposure to a pathogen culminated in one of the great milestones of medicine.
Smallpox was a highly prevalent disease that posed a major public health problem. It was widespread across much of the world, affected people of all ages and social classes, and caused high mortality rates (between 30% and 60% among the unvaccinated).
In 1796, the English scientist Edward Jenner inaugurated modern vaccination by demonstrating with practical evidence that cowpox protected against human smallpox. His discovery, the first to be scientifically documented, was based on various earlier inoculation experiments carried out in England by Lady Mary Wortley Montagu and subsequently by Benjamin Jesty using cowpox, as well as on Jenner’s own rural observation that milkmaids who contracted cowpox did not develop smallpox.
Thus emerged one of the greatest advances in the history of public health, even without any understanding of the existence of microorganisms, cells or internal defences. Immunology began as a discipline operating without knowledge of the underlying mechanisms, guided instead by evidence and results. The vaccine worked, even though no-one yet knew why. The impact of this discovery was both enormous and immediate.
Almost a century after Jenner’s experiments, the chemist Louis Pasteur began his work on vaccines. The success of Pasteur’s vaccinations using attenuated viruses made him a national hero and facilitated the creation of a number of vaccine institutes. Today, the Pasteur Institute is a leading centre for biomedical research and forms part of a network of 30 Pasteur Institutes across 27 countries on five continents.
Hygiene, infection and scientific common sense
While the development of vaccines was advancing to protect against other diseases, another silent revolution was taking shape.
In the 1850s, the English nurse Florence Nightingale observed that cleanliness, ventilation and order drastically reduced mortality in hospitals. The cause was unknown, but the practice saved lives.
In the mid-19th century, improvements in hospital hygiene and the introduction of handwashing significantly reduced deaths. Once again, medicine was facing a revolution that was taking place without understanding the mechanism behind it. It was not until 1876 that the German physician and microbiologist Robert Koch identified the anthrax bacillus and definitively demonstrated that microorganisms cause disease. Suddenly, everything made sense: hygiene was not only important for maintaining order, but above all it was a powerful barrier against invisible enemies. For the first time, the concept of a pathogen was described as an attacking enemy of the body, but it still remained to be understood who its defender was.
An invisible army within the body
The major questions of medicine then changed direction. It was no longer just a matter of identifying what made us ill, but of discovering what weapons the body possessed to defend itself, where they were and how they worked.
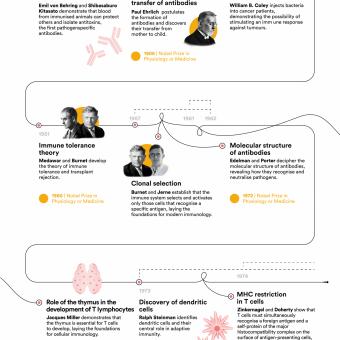
The development of new technologies such as the electron microscope and the refinement of cell culture techniques made it possible to discover that the body is populated by specialised cells. At the end of the 19th century, the microbiologist Ilya Ilyich Mechnikov discovered that some of these cells were capable of engulfing pathogens, a process he named phagocytosis. Other specialised cells were able to recognise infected cells and eliminate them, or even produce substances that neutralised the infection of other cells.
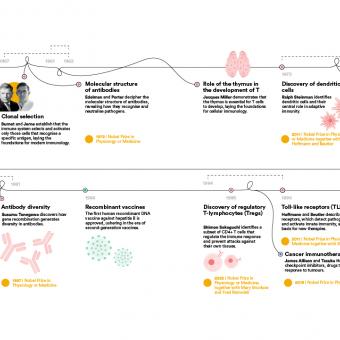
Gradually, what we now know as the immune system began to be described and understood: the set of cells, tissues and organs that protect the body from foreign substances and harmful cells. The immune system therefore comprises a wide range of cell types, such as T and B lymphocytes, neutrophils and dendritic cells, etc., as well as organs such as the bone marrow, thymus and spleen, and molecules such as antibodies.
A key feature of this system is its capacity for anticipation. The body already possesses a broad repertoire of lymphocytes with different receptors capable of recognising a wide variety of foreign elements. When these appear, the lymphocytes that recognise them are able to produce specific antibodies against them. The immune system functions as if it possessed many keys, ready to open an infinite number of locks.
Memory: the true learning of the immune system
When one of the lymphocytes in this prepared army finds its “lock”, the immune response against what must be destroyed is activated. The activated lymphocyte begins to multiply (it is said to clone itself) in order to create a large number of specialised soldiers. This new specialised army divides into those that take part in eliminating foreign elements and those that produce antibodies to block them.
When the battle ends, not everything disappears. A few memory cells remain, trained and ready to respond more rapidly if “the enemy” returns. These are the cells scientists aim to generate with each vaccine. In addition, in some cases long-lived plasma cells remain, capable of continuing to produce antibodies for years. Inducing this type of long-lasting protection is also one of the objectives of vaccines.
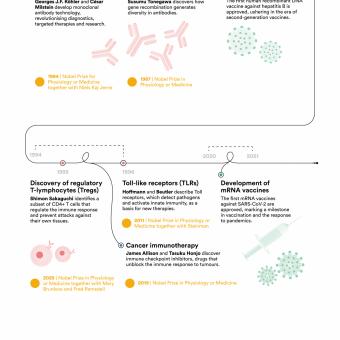
This principle, established during the 20th century with theories such as clonal selection, initiated with the studies of Frank Macfarlane Burnet and the theoretical work of Niels Kaj Jerne, both immunologists, explains why an infection or vaccination can provide protection for years or even decades.
When the enemy is inside
The immune system does not only deal with external infections. The body is also prepared to defend us against the emergence of malignant cells. This idea, now central to oncology, has a surprising origin.
At the end of the 19th century, the American physician William B. Coley observed that a patient with a tumour improved after a local infection. Thus, immunotherapy was born, once again from an observation whose underlying mechanisms were not yet understood. Today we know that behind this surprising phenomenon was the immune system itself, with lymphocytes specifically attacking tumour cells.
The problem is that cancer holds an advantage over pathogens. Tumour cells belong to the body itself, and for that reason they often go unnoticed. “The immune system has had to evolve to be very precise in what it does, attacking infections such as viruses or bacteria, but tolerating normal tissues. For that reason, the immune system has developed a series of checkpoints to activate or deactivate itself when necessary. Cancer has learned from this and tries to hide by exploiting the immune system’s negative controls,” explains Antoni Ribas, chairman of the Scientific Advisory Board of the CaixaResearch Institute.
The discovery of immune checkpoint inhibitors at the end of the 20th century by the immunologists James Allison and Tasuku Honjo made it possible to unblock that response and demonstrated that the immune system can be reactivated to fight advanced tumours. It was a revolution in cancer immunotherapy that earned them the Nobel Prize in Physiology or Medicine in 2019.
The delicate balance between defence and tolerance
This same system of control explains other phenomena. The immune system’s ability to distinguish between what belongs to our body and what does not lies, for example, behind the rejection of transplanted organs. In addition, when tolerance to the body’s own tissues fails, what are known as autoimmune diseases can develop.
“At a given moment, the immune system can function inappropriately. This can manifest as an exaggerated response to harmless antigens, which occurs in allergic diseases, or as a loss of tolerance to self-antigens, leading to autoimmune diseases,” explains Josep Tabernero, chairman of the Scientific Committee of the CaixaResearch Institute.
Autoimmunity is not a single disease but a group of disorders. It can affect different organs, its triggers combine genetic and environmental factors, and it can turn a temporary immune response into a chronic problem.
“Autoimmune responses include multiple conditions, such as multiple sclerosis, when the immune system damages certain components of the brain; diabetes, when it damages the pancreas; rheumatoid arthritis, when it affects joint structures; or psoriasis, in the case of the skin,” notes Gabriel Rabinovich, principal investigator at the CaixaResearch Institute.
Understanding how immune tolerance is maintained, and why it is sometimes lost, is one of the major current challenges in immunology.
Precision: the new language of immunology
Today, the rules of immunology are fairly well understood, but not completely. We know that some patients respond to a therapy and then stop doing so. Solving this puzzle is key to the future of medicine.
“Much of the research focuses precisely on understanding why this happens: why some people respond favourably and others do not, and whether the mechanisms are general or specific to each disease and each individual. Precision medicine aims to understand these individual and characteristic mechanisms so that they can be addressed in a personalised way, either in specific groups or at the individual level,” emphasises Dr Rabinovich.
“There is increasing evidence of the importance of an individual’s baseline immune state, since it can predict and determine how we will respond to vaccines, infections and other diseases,” explains Gemma Moncunill, principal investigator at the CaixaResearch Institute. The baseline state describes the configuration and functioning of an individual’s immune system under normal conditions. This state depends on genetic factors but also on environmental factors such as pollution, lifestyle and a person’s previous infections.
Looking at health through a new lens
Today we understand better the boundaries between the body, microorganisms and defences, but there is still room to refine, personalise and maximise therapeutic benefit. The current questions are no longer limited to how we defend ourselves, but also concern how to modulate, train and support the immune system throughout life. For this reason, the CaixaResearch Institute aims to place immunology at the centre of health, not only as a scientific ambition but also as a necessity in order to respond to the major medical challenges of the present and the future.